The PantId
Introduction—The Pantid®
Pantids® are specifically designed protein therapeutics than can (1) neutralize autoantibodies produced by B cells, (2) kill autoreactive B cells, and for, certain checkpoint inhibitor Pantids®, establish tolerogenesis, mitigating autoimmunity broadly.
While the precise initiating trigger for autoimmune disease development is not typically known, the autoantigen targets for specific autoimmune diseases have been identified in most diseases; collagen, for instance, is an autoantigen in rheumatoid arthritis that is bound by anti-collagen autoantibodies. Similarly, for the immune-related adverse events (irAEs) that develop after cancer immunotherapy, the targets are CTLA-4, PD-1, and PD-L1 proteins. During the design of Pantids®, the protein sequence of the autoantigen is literally copied into the Pantid® scaffold, and it is this autoantigen sequence that determines the specificity of the Pantid® as a therapeutic. As shown in Figure 1, the autoantigen sequence comprises the “binding domain” of the Pantid®. The “killing domain” of the Pantids® are cell-killing (i.e., cytotoxic) protein portions that are fused to the autoantigen “binding domain.” Because each autoimmune disease is characterized by a distinct and limited set of autoantigens, each Pantid®—containing these autoantigen sequences—is necessarily a specific therapeutic for that autoimmune disease. As such, there is no one “Pantid®” therapeutic, but rather a catalog of therapeutics, each of which is tailored for a specific patient or a specific autoimmune disease.
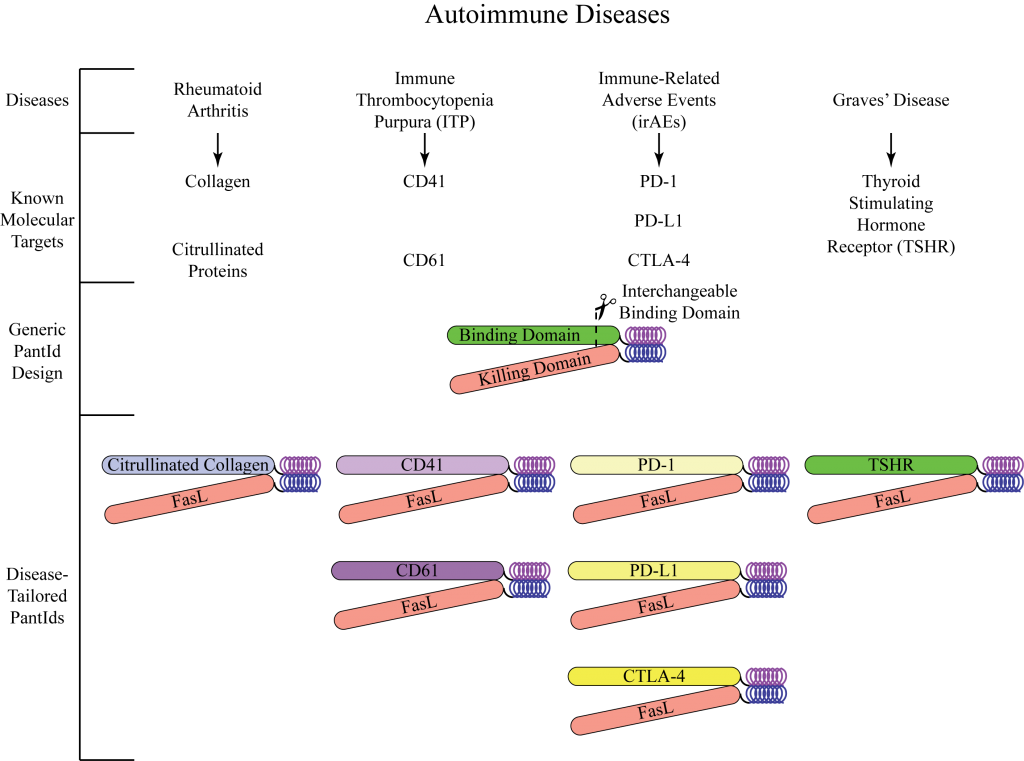
Figure 1: The Pantid® binding domain is interchangeable, allowing any autoimmune disease to be targeted.
PantIds® have three mechanisms of action
Neutralization
In many autoimmune diseases, autoreactive B cells produce soluble antibodies that are secreted into the surrounding tissues and systemic circulation. These autoantibodies can bind to their cognate autoantigens (the proteins, carbohydrates, or DNA they bind to), resulting in tissue dysregulation. When antibodies bind to antigen, they can indirectly induce the following: cell death through immune cells, particularly macrophages and NK cells; inflammation through eosinophils, macrophages, and neutrophils; and cell lysis through the complement system after a protein complex (i.e. the membrane attack complex) is formed. When normal tissues are bound by autoantibodies, any of these pathways can result in tissue damage and loss of function over time. Additionally, for soluble autoantigens, which often serve intercellular signaling functions, binding to autoantibodies can effectively prevent this intercellular communication, disrupting tissue functionality. In the special case of IRAEs, therapeutic antibodies in checkpoint inhibitor cancer immunotherapy hyper-stimulate the immune system through derepression, resulting in autoimmune diseases.
PantIds®, being comprised partly of autoantigen sequences, can bind to autoantibodies on their antigen-binding domains. This precludes further binding to autoantigen, as the antigen-binding sites are occupied by PantIds®, and this process is referred to as ”neutralization.” Both the PantId® and autoantibody are rendered largely inert in this process, as soluble immune complexes, which are removed from circulation and degraded in the spleen and liver.
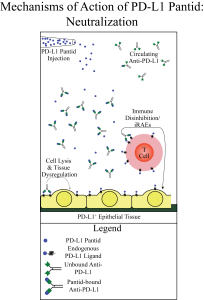
Figure 2: Pantids bind to circulating autoantibodies, neutralizing them and preventing tissue damage.
Cell Killing and Immunomodulation
As described above, PantIds® are a protein fusion of autoantigen and a cell-killing effector, which can directly kill cells through a regulated “cell-suicide” mechanism, denoted apoptosis. In addition to soluble autoantibodies, autoreactive B cells express a small amount of surface-bound (i.e. transmembrane) autoantibody, which is referred to as the B cell receptor (a.k.a. BCR). In instances where PantIds® bind to an autoreactive B cell through its BCR, the cell-killing effector binds to receptors on the B cell as well. Ultimately, the B cell will die though apoptosis in this process or be rendered inert (i.e. anergic). In most autoimmune diseases, the autoantibody repertoire is “oligoclonal,” which means that only a small number of autoreactive B cell clones are responsible for producing the entirety of autoantibodies in the patient. PantIds® are a unique therapeutic in that they can kill an entire B cell clone pool, which is broadly denoted “clonal deletion.” As only autoreactive B cells bind to the PantIds®, it is only these cells that are clonally deleted, leaving the remainder of the immune system unperturbed.
In addition to cell-killing effectors, our intellectual property includes PantIds® that have immunomodulatory effectors—proteins that do not kill immune cells, but rather prevent their pathological activation. This can be viewed as a “softer shutdown” to the aforementioned cell-killing mechanisms. We are currently characterizing and developing these PantIds® where direct cell killing could be problematic—in instances where the binding domain of the PantId® can also bind to tissues through a receptor

Figure 3: Pantids directly bind to autoreactive B cell BCRs, while the cell-killing or immunomodulatory domain results in clonal deletion or immunosuppression, respectively.
Tolerogenesis
Some of our PantIds® include native negative immune signaling molecules, called negative checkpoint receptors or ligands. Preliminary research indicates that these PantIds® directly modulate the immune system broadly and non-specifically (i.e. independent of any present or absent autoimmune disease), and in the absence of associated cell-killing or immunomodulatory effectors. These special PantIds® may ultimately be used as a general therapeutic in all autoimmune diseases, owing to their breadth of action. In this instance, PantIds® bind to immunoinhibitory receptors or ligands on immune cells, preventing their activation and rebalancing the immune system. This “rebalancing” process is referred as tolerogenesis, where autoreactive or hyperactive immune cells learn to “tolerate” the presence of cognate antigen, without causing tissue destruction.